Current Issue
Optimizing Gastrostomy Tube Placement in a Rural Infant Population
Jeffrey M. Oxford1, Meghan Groghan2, Elisabeth J. Leeflang2, Jason McKee3, Jessie R. Maxwell2,4*, Dawn Novak2
1University of New Mexico School of Medicine, Albuquerque, New Mexico
2Department of Pediatrics, University of New Mexico, Albuquerque, New Mexico
3Department of Surgery, University of New Mexico, Albuquerque, New Mexico
4Department of Neurosciences, University of New Mexico, Albuquerque, New Mexico
Corresponding author: Jessie Maxwell, MD, MBA, Department of Pediatrics, Division of Neonatology, University of New Mexico, MSC10 5590; 1 University of New Mexico, Albuquerque, New Mexico, 87131, United States, Phone: 505-272-0366, Fax: (505) 272-9643, Email: [email protected]
Received Date: January 08, 2026
Publication Date: February 07, 2026
Citation: Oxford MJ, et al. (2026). Optimizing Gastrostomy Tube Placement in a Rural Infant Population. Neonatal. 7(1):28.
Copyright: Oxford MJ, et al. © (2026).
ABSTRACT
Objectives: Feeding gastrostomy placement is a common surgical procedure in neonates. Predicting which infants require only short-term supported feedings could reduce the number of surgical gastrostomy tubes being placed and allow for the use of home nasogastric feedings. A home nasogastric feeding program in our rural population would require focusing on safety concerns. Aim: This study aims to better characterize infants requiring gastrostomy tube placement. Methods: A retrospective chart review was performed from January 1, 2018, through January 1, 2021, for infants in the Newborn Intensive Care Unit requiring gastrostomy tube placement before 12 months of age. Data on the feeding status at 2- and 6-months post-gastrostomy tube placement, the time to removal of gastrostomy tube, gastrostomy tube related complications, and other factors that might predict early removal of gastrostomy tube were collected. Results: Among the 104 infants included in the study, the mean time to reach full oral feeding volumes after gastrostomy tube placement was 18 months. Seven infants (6.7%) achieved full oral feeding volumes within 2 months, and 26 infants (25%) did so within 6 months. Conclusions: Given that the number of gastrostomy tubes used for less than 2 months was not significant, we do not recommend establishing a home nasogastric (NG) feeding protocol or program within our rural patient population.
Keywords: Gastrostomy Tubes, Rural Healthcare, Infants
INTRODUCTION
Optimal nutrition significantly impacts many aspects of infant development. Those with suboptimal nutrition may suffer from growth failure and metabolic disturbances that can persist into adulthood [1]. Poor nutrition among infants has also been shown to account for slow growth and increased risk of adverse complications such as necrotizing enterocolitis, late-onset sepsis, bronchopulmonary dysplasia, neurodevelopmental impairment, and death [2,3]. Additionally, infants born prematurely or with medical complications may have higher nutritional demands to maintain optimal growth, putting them at greater risk of poor nutrition [4]. This often necessitates supplementing oral nutritional intake with high-calorie foods or increasing the nutrient density of foods using high-energy fats, carbohydrates, and proteins.
Supplemental nutrition can be given to infants in many ways. Factors that influence the choice of administration include the expected duration of the need for supplemental nutrition, possible risk of aspiration, and the integrity of the upper gastrointestinal tract [5-7]. Enteral feeding is defined as nutrition given either by mouth or through a feeding tube, whereas parenteral nutrition refers to supplemental nutrition provided via a venous catheter into the bloodstream. If the infant is not able to safely or completely ingest oral enteral nutrition, it can be delivered via a nasogastric (NG) tube, gastrostomy tube (G-tube), gastro-jejunostomy tube, or jejunostomy tube. These methods may be indicated in infants with impaired swallowing or oral motor development, excessive metabolic demands, or impaired absorption or digestion, as well as those requiring certain support for respiration [8]. Enteral nutrition is preferred over parenteral nutrition in patients with a functioning gastrointestinal (GI) tract [5] due to improved absorption while avoiding complications of parenteral nutrition, including infection and cholestasis [9]. Intragastric feeding, when possible, is preferred as it activates the normal neurological and hormonal pathways in digestion and absorption, and is simpler than placing a post-pyloric tube [6]. As such, NG tubes are the most used feeding method in infants while hospitalized.
Once infants are medically stable, a safe home feeding plan must be considered. G-tube placement offers more stable and secure feeding access, reducing the risk of dislodgment (with potentially more emergent visits related to complications for dislodgment) after hospital discharge [10]. G-tubes can be hidden under clothing and don’t affect the child’s ability to vocalize, while NG tubes can be replaced safely by trained personnel or even family members but can require X-ray imaging to confirm placement. G-tube placement requires surgery, and thus has associated risks, including anesthetic complications, tube blockage, stoma site infection, and leakage [11]. In determining whether a G-tube or NG tube is most appropriate for a patient, the expected duration of use and the risks and benefits must be considered.
In a primarily rural population, there are additional important considerations. Distance to nearest medical center with X-ray, pediatric expertise, and appropriately sized tubes, reliable transportation and medical literacy along with family comfort are all key concerns in developing a safe feeding and discharge plan for these infants. Long-term NG feedings pose more challenges for families, especially with other children at home or working parents, as NG tubes require nearly constant attention to ensure correct placement. Whereas, once the GT tract is formed (~6-8 weeks), families can be confidently trained to replace the GT safely. Families should be counseled to understand the factors of NG versus GT feeds in a risk-benefit ratio, including aspiration, pneumonia, peritonitis, and even death in the event of feeding through an improperly positioned tube.
This study aimed to characterize infants requiring G-tube placement and determine the duration of use until full oral feeding volumes were reached. In infants who required the G-tube for a short period (<2 months), additional analyses were completed to determine if specific factors could be identified to allow for stratification of those who could potentially benefit from a home nasogastric tube program rather than needing a G-tube placed.
METHODS
Following Institutional Review Board approval at the University of New Mexico Research Protections Office, a retrospective chart review was completed from January 1, 2018, through January 1, 2021. Participants were identified using the Current Procedural Terminology (CPT) code 43653 (laparoscopic gastrostomy), after which additional inclusion and exclusion criteria were applied. Inclusion criteria included infants under one year of age admitted to the neonatal intensive care unit (NICU) who had post-discharge follow-up visits within the electronic medical system (EMR) for at least 2 additional months. Infants with incomplete records, no follow-up data available, or those who were wards of the state were excluded.
Once eligible participants were identified, a unique study code was applied for de-identification purposes, and study data was collected and managed using REDCap electronic data capture tools. Demographic data collected included infant sex, zip code of primary residence, gestational age at birth and type of caregiver at discharge. Information collected relating to the G-tube included diagnosis or specific indication for G-tube placement, age at time of surgery, percentage of oral feeds at time of surgery, weight at surgery, and swallow study findings (if the study had been completed). The presence of potential factors contributing to the need for a G-tube, such as congenital anomalies, intraventricular hemorrhage, hypoxic-ischemic encephalopathy, and intrauterine drug exposure were also recorded. Finally, data related to the outcome of the G-tube placement, such as known complications and how those complications were managed, time to full oral feeds after G-tube surgery, time from G-tube placement to hospital discharge, weight at hospital discharge, total length of hospital stay, and duration of G-tube use (when known) were analyzed. Any known emergency room, urgent care, or other clinic visits as well as any hospitalizations related to potential G-tube complications were noted. The European Society for Parenteral and Enteral Nutrition home enteral nutrition guideline recommends using NG feedings for a maximum of 4-6 weeks, with gastrostomy tubes recommended beyond that time [12]. Therefore, we categorized the duration of G-tube use into short-term (<2 months), moderate duration (≤2 to <6 months), longer term use (≥6 months) to help further distinguish potential situations in which a g-tube may or may not have been indicated.
Demographics were summarized using percentages for categorical variables and means with standard error of the means for continuous variables. A one-way analysis of variance (ANOVA) was used to compare the infants requiring a G-tube for 0 to <2 months (considered short term use), ≥2 to <6 months (considered moderate duration of use), and ≥6 months (considered longer term use) for continuous variables. A Chi-Square analysis was completed for categorical variables. A p-value of <0.05 was considered statistically significant.
RESULTS
A total of 3,322 infants were admitted to the University of New Mexico Hospital between January 1, 2018, and January 1, 2021. Of the 3,322 infants admitted, only 156 infants (4.7%) underwent G-tube placement surgery. For the purposes of this study, fifty-two infants were excluded for either lack of follow-up information (n=18), being admitted in a unit other than the NICU (n=22) or being wards of the state (n=12). Thus, 104 infants met inclusion and exclusion criteria (Figure 1).
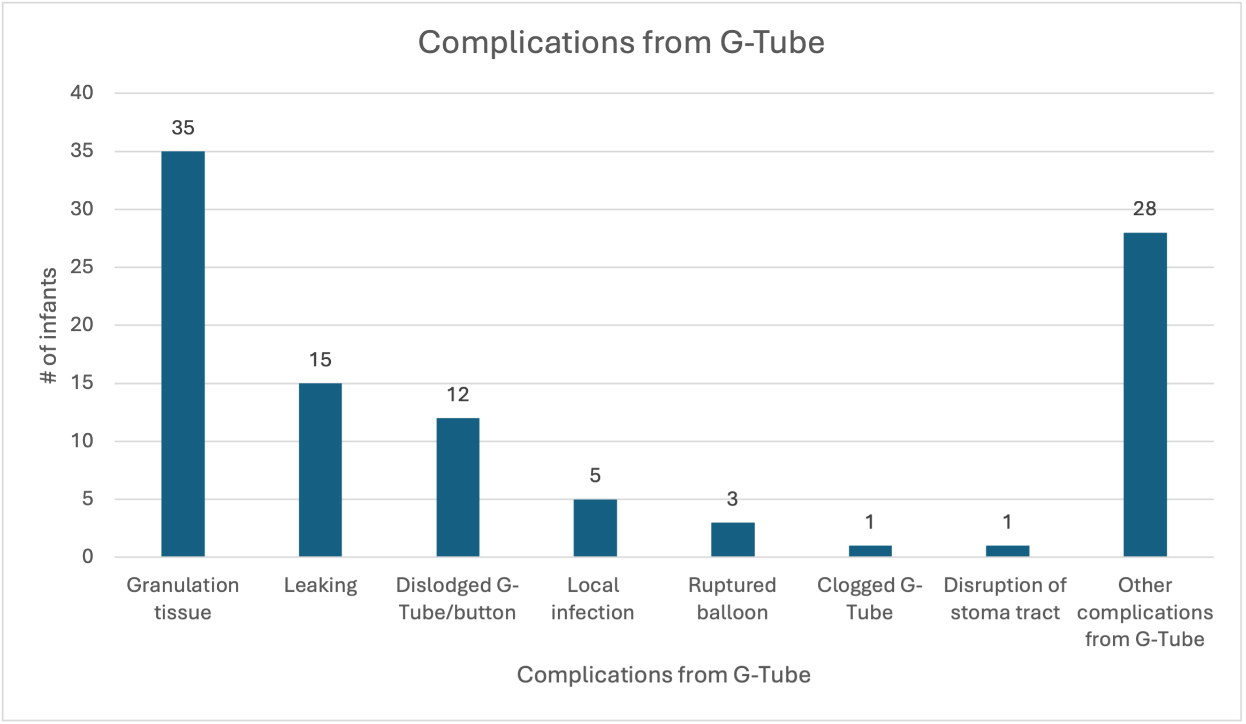
Figure 1: Complications After G-tube Placement
A number of complications occurred after G-tube placement. The most common complication noted was granulation tissue around the insertion site. More complex issues, such as disruption of the stoma tract and ruptured balloon, were far less common.
The average gestational age (GA) at birth for the 104 infants included was 35 weeks and 2 days (see Table 1 for Demographic Characteristics). Overall, there was similar number of female (n=45, 43.3%) and male (n=59, 56.7%) infants. Interestingly, most infants who required a G-tube during their hospitalization had congenital anomalies (n=73, 70.2%). Other factors that can contribute to the success of oral feeding were observed in a smaller number of infants, including intrauterine drug exposure (n=15, 14.4%), intraventricular hemorrhage (n=12, 11.5%), and hypoxic ischemic encephalopathy (n=6, 5.8%). A variety of syndromes, chromosomal abnormalities, and other physical anomalies were present within the population of infants who required G-tube placement. The syndromes or associations diagnosed in this population included Cri du Chat, Prader Willi, Potacki-Lupski, Pierre-Robin, Stickler, DiGeorge, Walker-Wardburg, Neurofibromatosis Type I, Phelan-McDermid, Pallister-Killian, Dandy Walker, Joubert's, and Angelman. Chromosomal abnormalities diagnosed included Trisomy 21, Trisomy 18, Trisomy 13, CYP24A1 variant, deletion of Yq11.23 AZFC region, duplication of 2q13, duplication of 15q11.2 and 3p26.3, duplication of 3q29, partial chromosome 18 deletion, 1q24-25 microdeletion, 15q13.3 and Xq22.3 duplication, and other variants of unknown significance. Physical anomalies not diagnosed as part of a syndrome included cardiac (left ventricular hypertrophy, right ventricular hypertrophy, interrupted inferior vena cava, large ventricular septal defect, Tetralogy of Fallot), neurologic (myelomeningocele, ventriculomegaly, absent septum pellucidum, septo-optic dysplasia, tethered cord, holoprosencephaly, agenesis of the corpus callosum), renal (posterior urethral valves, bilateral severe hydronephrosis, ureteropelvic junction obstruction, congenital hydronephrosis), gastrointestinal (duodenal atresia, colonic agenesis, ileal atresia, imperforate anus), pulmonary (congenital diaphragmatic hernia, laryngomalacia, tracheoesophageal fistula, congenital pulmonary hypoplasia, obstructive sleep apnea, severe tracheomalacia), and skeletal / facial (vertebral anomalies, skeletal anomalies, craniosynostosis, submucosal cleft palate, micrognathia, mesenchymal facial tumor, choanal atresia). While many of these defects may not have directly contributed to the need for a G-tube, they do highlight the commonality of anomalies present within this population.
|
Characteristic |
Mean ± SEM |
|
Average gestational age at birth |
35.2 ± 5.3 |
|
|
n (%) |
|
Sex |
|
|
Male |
59 (56.7) |
|
Female |
45 (43.3) |
|
Congenital anomalies |
|
|
Syndrome / Association |
12 (11.5) |
|
Chromosomal Abnormality |
16 (15.4) |
|
Physical Anomaly |
45 (43.3) |
|
Intraventricular hemorrhage (IVH) |
|
|
Grade I IVH |
2 (1.9) |
|
Grade II IVH |
5 (4.8) |
|
Grade III IVH |
1 (1.0) |
|
Grade IV IVH |
4 (3.8) |
|
Hypoxic-Ischemic Encephalopathy (HIE) |
|
|
Mild HIE |
1 (1.0) |
|
Moderate HIE |
1 (1.0) |
|
Severe HIE |
4 (3.8) |
|
Intrauterine drug exposure (IUDE)* |
|
|
Presence of IUDE |
15 (14.4) |
|
Methamphetamines |
8 (7.7) |
|
Cannabinoids |
7 (6.7) |
|
Cocaine |
3 (2.9) |
|
Opiates |
2 (1.9) |
|
Fentanyl |
1 (1.0) |
|
Other Substance |
4 (3.8) |
SEM: Standard Error of the Mean.
*The subcategories of substances are not mutually exclusive; an infant may have more than one substance exposure included.
The average time for infants to reach full oral feeding volume goals following placement of gastrostomy tube was 18.4 months, with a range of 1 to 56 months. Infants were considered to require the G-tube for a short period if they reached full oral feeding volumes within 2 months; 7 infants (6.7%) met this goal. The majority of infants required the G-tube for longer than 6 months (n=78, 75.0%), with 19 infants (18.3%) reaching full oral feeding volumes between 2-6 months after gastrostomy tube placement. Of the 7 infants requiring the G-tube for the shortest duration, the gestational age was similar to the general cohort (35 weeks and 4 days), but overall, there were fewer other factors present (Table 2). None of the 7 infants had intraventricular hemorrhage or a diagnosis of hypoxic ischemic encephalopathy. Only 2 infants were noted to have congenital anomalies (n=2, 28.6%). Of the 19 infants who required the G-tube for 2-6 months, more co-diagnoses were present, including congenital anomalies (n=7, 36.8%), intraventricular hemorrhage (n=3, 15.8%), and hypoxic ischemic encephalopathy (n=1, 5.3%). When evaluating the infants who required the G-tube for 6 months or more, congenital anomalies became statistically more common (n=64, 82%, p<0.001), while a diagnosis of hypoxic ischemia encephalopathy increased slightly (n=5, 6.4%; Table 2). Interestingly, while the mean gestational age at birth and mean gestational age at the time of G-tube placement surgery were similar amongst all groups, infants who required the G-tube for ≥6 months were significantly smaller weight at the time of surgery (p<0.05; Table 3).
|
Full oral feeding <2 months (n=7) |
Full oral feeding 2-6 months (n=19) |
Full oral feeding ≥6 months (n=78) |
P-Value |
|
|
Characteristic |
Mean ± SEM |
Mean ± SEM |
Mean ± SEM |
|
|
Gestational age at birth (weeks) |
35.5 ± 3.4 |
33.3 ± 1.3 |
35.7 ± 1.9 |
0.82* |
|
Age at surgery (days) |
59.6 ± 11.3 |
71.0 ± 9.1 |
59.0 ± 4.8 |
0.52* |
|
Weight at surgery (kg) |
4.7 ± 0.3 |
4.2 ± 0.2 |
3.9 ± 0.1 |
0.04* |
|
|
n (%) |
n (%) |
n (%) |
|
|
Sex |
|
|
|
0.21^ |
|
Male |
6 (85.7) |
9 (47.4) |
44 (56.4) |
|
|
Female |
1 (14.3) |
10 (52.6) |
34 (43.6) |
|
|
Congenital Anomaly |
2 (28.6) |
7 (36.8) |
64 (82.1) |
<0.001^ |
|
Syndrome / Association |
0 (0.0) |
1 (5.3) |
11 (14,1) |
|
|
Chromosomal Abnormality |
0 (0.0) |
1 (5.3) |
15 (19.2) |
|
|
Physical Anomaly |
2 (28.6) |
5 (26.3) |
38 (48.7) |
|
|
No Congenital Anomaly |
5 (71.4) |
12 (63.2) |
14 (17.9) |
|
|
Intraventricular Hemorrhage (IVH) |
0 (0.0) |
3 (15.8) |
9 (11.5) |
0.53^ |
|
Grade I IVH |
0 (0.0) |
1 (5.3) |
1 (1.3) |
|
|
Grade II IVH |
0 (0.0) |
0 (0.0) |
5 (6.4) |
|
|
Grade III IVH |
0 (0.0) |
1 (5.3) |
0 (0.0) |
|
|
Grade IV IVH |
0 (0.0) |
1 (5.3) |
3 (3.8) |
|
|
No IVH |
7 (100.0) |
16 (84.2) |
69 (88.5) |
|
|
Hypoxic Ischemic Encephalopathy (HIE) |
0 (0.0) |
1 (5.3) |
5 (6.4) |
0.57^ |
|
Mild HIE |
0 (0.0) |
0 (0.0) |
1 (1.3) |
|
|
Moderate HIE |
0 (0.0) |
1 (5.3) |
0 (0.0) |
|
|
Severe HIE |
0 (0.0) |
0 (0.0) |
4 (5.1) |
|
|
No HIE |
7 (100.0) |
18 (94.7) |
73 (93.6) |
|
SEM: Standard Error of the Mean; kg: kilogram; ANOVA: One-way analysis of variance.
*One-way ANOVA analysis used; ^Chi-square analysis used
Table 3: Clinical Information at Time of G-tube Placement Based on G-tube Duration
|
Total (N=104) |
Full oral feeding <2 months (n=7) |
Full oral feeding 2-6 months (n=19) |
Full oral feeding ≥6 months (n=78) |
P-Value |
|
|
|
Mean ± SEM |
Mean ± SEM |
Mean ± SEM |
Mean ± SEM |
|
|
Percentage of oral feeds at time of surgery (%) |
14.8 ± 2.2 |
38.3 ± 5.8 |
16.5 ± 4.8 |
12.0 ± 2.3 |
0.005* |
|
Time from surgery to discharge (days) |
22.6 ± 4.1 |
7.4 ± 1.0 |
10.5 ± 9.6 |
28.2 ± 5.3 |
0.19* |
|
Length of hospital stay (days) |
83.0 ± 6.5 |
67.9 ± 11.4 |
81.5 ± 15.3 |
87.8 ± 8.5 |
0.75* |
|
Weight at discharge (kg) |
4.5 ± 0.1 |
4.9 ± 0.3 |
4.4 ± 0.3 |
4.5 ± 0.2 |
0.72* |
|
Duration of G-tube (months) |
22.3 ±1.4 |
7.1 ± 1.4 |
10.7 ± 3.2 |
26.2 ± 1.5 |
<0.0001* |
SEM: Standard Error of the Mean; kg: kilogram.
*One-way ANOVA analysis used
Overall, the average percentage of oral feeds for all infants at the time of the G-tube placement was 14.8%. Infants who required the G-tube for <2 months were taking a significantly higher volume of oral feeds (38.3%) compared to either those who required the G-tube for 2-6 months (16.5%) or more than 6 months (12.0%; p<0.01; Table 3). While infants who required the G-tube for the longest duration were the smallest at the time of surgical placement, all infants had a similar weight at time of discharge (p=0.72). No differences were observed between the days from the surgical G-tube placement to discharge or the total length of hospital stay.
There was no significant difference between the groups as to utilization of either outpatient or inpatient medical care related to the G-tube (Table 4). The most common complications that occurred related to the G-tube included granulation tissue around the insertion site and leaking. Complex issues, such as ruptured balloon and disrupted stoma tract, were the least commonly observed complications (Figure 1).
Table 4: Medical Care Utilization After G-tube Placement
|
Total (N=104) |
Full oral feeding <2 months (n=7) |
Full oral feeding 2-6 months (n=19) |
Full oral feeding ≥6 months (n=78) |
P-Value |
|
|
|
Mean ± SEM |
Mean ± SEM |
Mean ± SEM |
Mean ± SEM |
|
|
Outpatient Medical Care Utilization Related to G-tube |
1.32 ± 0.16 |
0.29 ± 0.29 |
0.89 ± 0.38 |
1.51 ± 0.20 |
0.10* |
|
Inpatient Medical Care Utilization Related to G-Tube |
0.08 ± 0.03 |
0.14 ± 0.14 |
0.05 ± 0.06 |
0.08 ± 0.03 |
0.75* |
SEM: Standard Error of the Mean.
*One-way ANOVA analysis used
DISCUSSION
Interestingly, a minority of patients with G-tube placement reached full oral feeding volumes within two months of surgery, indicating that overall, the placement of G-tubes appears to be appropriate within our population. Prior studies suggest that infants who do not reach full oral feedings within 60 days from hospital discharge are likely to require tube feeds for an extended period and would benefit from a surgically placed G-tube [13]. Generally, our practice is to allow the infant to reach their estimated delivery date or demonstrate failure to adequately tolerate taking sufficient volume by mouth at 42 weeks corrected gestational age before discussing a G-tube placement. Therefore, even if a home NG feeding plan could have been safely used, only 7 infants (less than 10%) may have avoided the G-tube surgery.
While literature widely supports that “short-term” feeding may be administered via a NG or orogastric (OG) tube and “long-term” feeding should be administered through a surgically placed G-tube [14], the timing of “short-term” and “long-term” are not well characterized. The infants with “short-term” use in this study were those requiring a G-tube for less than 2 months to be in alignment with the European Society for Parental and Enteral Nutrition home enteral nutrition guidelines. Additionally, individual diagnoses, and home environment including caregiver training, capability, and time to the nearest medical center, should be carefully evaluated in determining the best treatment option for each individual [5,6,10,15-17].
A survey in one rural area of New Mexico showed that 84% of individuals reported using a bus system to access health services, but that many challenges are present for public transportation. Within rural New Mexico, individuals are often required to travel long distances with poor road conditions, complicating the ability for public transportation to be effective, if it even exists in the region [18]. Even within the largest metropolitan area of New Mexico, primary healthcare access was low amongst those who relied heavily on public transportation [19]. Further complicating the decision for G-tube placement is that the duration of enteral nutrition support depends on many factors and is often difficult to predict.
The most common indication for supplemental nutrition via a surgically placed device in infants is oropharyngeal dysphagia [20,21]. Underlying causes of oropharyngeal dysphagia vary widely, and can include neurodevelopmental impairment due to hypoxia, intracranial hemorrhage, congenital central nervous system impairment, hypotonia of muscular, metabolic or neurologic origin, and anomalies of mouth, pharynx and larynx, among other etiologies [22-25]. It was not surprising to observe more infants with congenital anomalies requiring longer use of a G-tube in our study. A large European cohort study found that children with congenital anomalies were 80 times more likely to need a G-tube compared to children without congenital anomalies [26].
Certain factors can improve the likelihood of successful oral feeding. Infants with a consistent feeding environment often have improvement in their feeding abilities [15]. Although several studies suggest that earlier discharge with home gavage feeding may be a preferrable alternative while also reducing the cost of hospital care [27,28], multiple risks are known. Testing the gastric pH prior to using an NG is intended to confirm the placement of the NG tube, however medications or oral consumption prior to pH testing can alter these results [29]. Many complications can occur due to misplacement of an NG, including esophageal or pulmonary perforation as well as pleural injury, pneumothorax, tracheobronchial aspiration, and infection [30]. Complications of long-term NG tube placement can include irritation and/or erosion of the gastric lining, electrolyte imbalances, ulceration, and gastrointestinal bleeding [30]. New Mexico is the 5th largest state by area in the United States, but there are fewer than 6 people per square mile of the state, and the majority of the roads are unpaved (57%) [31]. This results in a delay in accessing health care, which could result in life threatening complications for home NG use.
Thus, caregivers should understand the complications associated with the various modes of assisted nutrition in the home setting. It is vital to have the proper resources for continued nutritional support after discharge from the hospital [6]. While NG tube placement may be preferable for a specific subpopulation of infants who require assisted nutrition at home, sufficient caregiver support and access to healthcare facilities must be demonstrated.
Overall, this study has many strengths, including a unique and diverse population in a rural state. Additionally, the inclusion of infants identified with congenital anomalies was robust. Given that this was a chart review, there are limitations in the data collected. A future prospective study could include all infants requiring G-tube placement (rather than those limited to follow up within 1 medical system), data on discussions with caregivers about nasogastric and gastric feeding programs, and information regarding discharge environment and the impact on the discharge feeding plan.
CONCLUSION
Among the 104 infants included in the study, the mean time to reach full oral feeding volumes after gastrostomy tube placement was 18 months. Most infants (n=78, 75%) required long-term gastric feedings >6 months after G-tube placement. Seven infants (6.7%) achieved full oral feeding volumes within 2 months, and 19 infants (18.3%) did so within 6 months. This study showed long-term gastric feeding was significantly associated with congenital anomalies and smaller weight and should be taken into consideration for home feeding plans. Currently, no home nasogastric feeding protocol is in use at our institution. Given that the number of gastrostomy tubes used for less than 2 months was not significant, we do not recommend establishing a new home nasogastric feeding protocol or program within our rural patient population at this time.
ACKNOWLEDGEMENTS
This study was supported by the Department of Pediatrics, Division of Neonatology.
FUNDING
No funding was received for the conduct of this study or the preparation of this manuscript.
CONFLICTS OF INTEREST
The authors declare that they have no conflicts of interest related to this study.
REFERENCES
- Patel JK, Rouster AS. (2025). Infant Nutrition Requirements and Options. StatPearls. Treasure Island (FL).
- Salas AA, Travers CP. (2023). The Practice of Enteral Nutrition: Clinical Evidence for Feeding Protocols. Clinics in Perinatology. 50(3):607-623.
- Ehrenkranz RA, Das A, Wrage LA, Poindexter BB, Higgins RD, Stoll BJ, et al. (2011). Early nutrition mediates the influence of severity of illness on extremely LBW infants. Pediatric Research. 69(6):522-529.
- Salvatori G, Martini L, The Study Group On Neonatal N, Gastroenterology-Italian Society Of Neonatology OBO. (2020). Complementary Feeding in the Preterm Infants: Summary of Available Macronutrient Intakes and Requirements. Nutrients. 12(12):3696.
- Braegger C, Decsi T, Dias JA, Hartman C, Kolacek S, Koletzko B, et al. (2010). Practical approach to paediatric enteral nutrition: a comment by the ESPGHAN committee on nutrition. Journal of Pediatric Gastroenterology and Nutrition. 51(1):110-122.
- Bering J, DiBaise JK. (2022). Home Parenteral and Enteral Nutrition. Nutrients. 14(13):2558.
- White BR, Zhang C, Presson AP, Friddle K, DiGeronimo R. (2019). Prevalence and outcomes for assisted home feeding in medically complex neonates. Journal of Pediatric Surgery. 54(3):465-470.
- Marchand V, Baker SS, Baker RD. (1998). Enteral nutrition in the pediatric population. Gastrointestinal Endoscopy Clinics of North America. 8(3):669-703.
- Kim AY, Lim RK, Han YM, Park KH, Byun SY. (2016). Parenteral Nutrition-Associated Cholestasis in Very Low Birth Weight Infants: A Single Center Experience. Pediatric Gastroenterology, Hepatology & Nutrition. 19(1):61-70.
- Khalil ST, Uhing MR, Duesing L, Visotcky A, Tarima S, Nghiem-Rao TH. (2017). Outcomes of Infants With Home Tube Feeding: Comparing Nasogastric vs Gastrostomy Tubes. JPEN. Journal of Parenteral and Enteral Nutrition.41(8):1380-1385.
- Lee J, Spratling R. (2014). Care of gastrostomy feeding tube in children with developmental disabilities. Rehabilitation Nursing. 39(5):221-224.
- Bischoff SC, Austin P, Boeykens K, Chourdakis M, Cuerda C, Jonkers-Schuitema C, et al. (2022). ESPEN practical guideline: Home enteral nutrition. Clinical Nutrition. 41(2):468-488.
- Williams SL, Popowics NM, Tadesse DG, Poindexter BB, Merhar SL. (2019). Tube feeding outcomes of infants in a Level IV NICU. Journal of Perinatology. 39(10):1406-1410.
- Holmes S. (2004). Enteral feeding and percutaneous endoscopic gastrostomy. Nursing standard. 18(20):41-43.
- Lagatta JM, Uhing M, Acharya K, Lavoie J, Rholl E, Malin K, et al. (2021). Actual and Potential Impact of a Home Nasogastric Tube Feeding Program for Infants Whose Neonatal Intensive Care Unit Discharge Is Affected by Delayed Oral Feedings. The Journal of Pediatrics. 234:38-45.e2.
- McAndrew S, Acharya K, Westerdahl J, Brousseau DC, Panepinto JA, Simpson P, et al. (2019). A Prospective Study of Parent Health-Related Quality of Life before and after Discharge from the Neonatal Intensive Care Unit. J Pediatr. 213:38-45.e3.
- Pedron-Giner C, Calderon C, Martinez-Zazo A, Canedo Villaroya E, Malillos Gonzalez P, Sesmero-Lillo MA. (2012). Home enteral nutrition in children: a 10 year experience with 304 pediatric patients. Nutricio?n Hospitalaria. 27(5):1444-1450.
- Del Rio M, Hargrove WL, Tomaka J, Korc M. Transportation Matters: A Health Impact Assessment in Rural New Mexico. (2017). International Journal of Environmental Research and Public Health. 14(6):629.
- Lardier DT, Jr., Blackwell MA, Beene D, Lin Y. (2023). Social Vulnerabilities and Spatial Access to Primary Healthcare through Car and Public Transportation System in the Albuquerque, NM, Metropolitan Area: Assessing Disparities through GIS and Multilevel Modeling. Journal of Urban Health. 100(1):88-102.
- Ramelli GP, Aloysius A, King C, Davis T, Muntoni F. (2007). Gastrostomy placement in paediatric patients with neuromuscular disorders: indications and outcome. Developmental medicine and child neurology. 49(5):367-371.
- Di Leo G, Pascolo P, Hamadeh K, Trombetta A, Ghirardo S, et al. (2019). Gastrostomy Placement and Management in Children: A Single-Center Experience. Nutrients. 11(7):1555.
- Law JB, Wood TR, Gogcu S, Comstock BA, Dighe M, Perez K, et al. (2021). Intracranial Hemorrhage and 2-Year Neurodevelopmental Outcomes in Infants Born Extremely Preterm. The Journal of pediatrics. 238:124-134.e10.
- Miguel PM, Silveira PP. (2020). Neonatal Hypoxia Ischemia and Individual Differences in Neurodevelopmental Outcomes. JAMA Pediatrics. 174(8):803.
- Ruecker M, Zepharovich K, Zorowka P, Matzak H, Kofler M, Saltuari L. (2018). Dysphagia in cerebral hypoxia. NeuroRehabilitation. 43(4):387-393.
- LaMantia AS, Moody SA, Maynard TM, Karpinski BA, Zohn IE, Mendelowitz D, et al. (2016) Hard to swallow: Developmental biological insights into pediatric dysphagia. Developmental Biology. 409(2):329-342.
- Garne E, Tan J, Loane M, Baldacci S, Ballardini E, Brigden J, et al. (2022). Gastrostomy and congenital anomalies: a European population-based study. BMJ Paediatrics Open. 6(1):e001526.
- Berman L, Baird R, Sant'Anna A, Rosen R, Petrini M, Cellucci M, et al. (2022). Gastrostomy Tube Use in Pediatrics: A Systematic Review. Pediatrics. 149(6):e2021055213.
- Sturm LD. Implementation and evaluation of a home gavage program for preterm infants. (2005). Neonatal Netw. 24(4):21-25.
- Alliance SCF, Breik L, Chaffey E, Chan K, Coy J, Crockett N, et al. (2025). Gold Standard Home Nasogastric Tube Feeding for Infants and Children. Journal of Paediatrics and Child Health. 61(7):1006-1013.
- Sigmon DF, An J. (2025). Nasogastric Tube. StatPearls. Treasure Island (FL).
- Bureau USC. (2022). [Available from: https://data.census.gov/all?q=new+mexico].
 Abstract
Abstract  PDF
PDF