Current Issue
Neonatal Hypernatremic Dehydration: A Retrospective Study of Risk Factors and Treatment
Patricia Campos Magallón1*, María De Pablo De Las Heras1, Ingrid Royo Sesma1, Sara Pasamón García1, Inés Esteban Diez1, Ana Ibiricu Lecumberri1, Bibiana Riaño Méndez1
1Hospital San Pedro. Calle Piqueras, Number 98, Zip Code 26006, Logroño (La Rioja), Spain
*Corresponding author: Patricia Campos Magallón, Calle Marqués de Larios, No. 30, 6ºD, Zip Code 26007, Logroño (La Rioja), Spain, Phone: +34 618016547, E-mail: [email protected]
Received Date: July 28, 2025
Publication Date: April 13, 2026
Citation: Magallón PC, et al. (2026). Neonatal Hypernatremic Dehydration: A Retrospective Study of Risk Factors and Treatment. Neonatal. 7(1):32.
Copyright: Magallón PC, et al. © (2026).
ABSTRACT
Neonatal hypernatremic dehydration is a potentially life-threatening condition that can cause significant neurological damage or death. The objective of this study is to determine its incidence in our setting, identify risk factors, and evaluate any statistical association between treatment and complications, aiming to generate new hypotheses to guide future research that could support standardized management protocols. This is a retrospective observational cohort study of newborns diagnosed with neonatal hypernatremic dehydration between May 2015 and January 2023 in a level III neonatal intensive care unit. Twenty-five patients were included. The main reason for seeking care was referral by another provider to the emergency department (36%). Mean weight loss relative to birth weight was 16%. Identified risk factors were primiparity (p=0.007), exclusive breastfeeding (p<0.001), and male sex (p=0.036). Sixteen percent developed complications. Significant associations were found between complications and initial volume expansion (p=0.007) and a sodium correction rate ≥ 0.5 mEq/L/h (p=0.041). A direct linear relationship was observed between infused intravenous volume and the rate of serum sodium decline (p<0.001, R²=0.733). Early follow-up, especially for high-risk neonates, is crucial to prevent or promptly detect hypernatremic dehydration. This study is limited by its small sample size, single-center design, and possible false positives due to multiple comparisons. Further research is needed to strengthen the evidence base. Hypotheses include evaluating whether restricting initial volume expansion to cases of shock reduces complications and whether initiating intravenous rehydration slowly, with gradual escalation and early combination with enteral feeds, reduces complications by avoiding serum sodium decreases greater than 0.5 mEq/L/h.
Keywords: Neonatal, Dehydration, Hypernatremia, Breastfeeding, Fluid Therapy
INTRODUCTION
Neonatal hypernatremic dehydration (NHD) is defined by serum sodium levels ≥150 mEq/L. It is a potentially severe condition that can lead to significant neurological sequelae and even death [1].
NHD occurs primarily as a result of insufficient breast milk intake combined with increased insensible water losses during the neonatal period. The reported incidence in population-based studies varies widely, ranging from 0.5 to 10.4 per 1,000 live newborns [2,3].
NHD is challenging to diagnose due to its presentation with nonspecific symptoms such as lethargy or irritability. Signs of dehydration may also be present, including dry mucous membranes, sunken fontanelle, poor skin turgor, signs of malnutrition, jaundice, fever, oliguria or anuria, and neurological alterations [4].
Described risk factors include weight loss >10% at maternity unit discharge, low birth weight, prematurity, exclusive breastfeeding, primiparity, early postpartum discharge, and concomitant neonatal jaundice [5].
The management of these patients remains controversial, with no established clinical practice guidelines. There is no consensus regarding the choice of fluids or the recommended volume for rehydration therapy in neonates requiring intravenous rehydration. The main points of agreement among published guidelines are the recognition of hypernatremia as a serious condition and the necessity to reduce serum sodium gradually, at a maximum rate of 0.5 mEq/L per hour (10–12 mEq/L of sodium per day) [6,7].
Most published studies agree that if the neonate is not in shock, rehydration can be performed enterally, using breast milk or a substitute [2].
Correction of hypernatremia is suggested to occur over 48 to 72 hours. The rate of correction depends on the severity of the disorder; the more severe the condition, the slower the correction should be [8].
Some complications secondary to NHD arise from cerebral edema formation, including intracranial hemorrhage, cerebral venous sinus thrombosis, and peripheral thrombosis of the renal vein. Extracerebral complications of hypernatremic dehydration encompass acute renal failure, elevated hepatic enzymes, metabolic acidosis, hyperbilirubinemia, hypoglycemia or hyperglycemia, and disseminated intravascular coagulation, among others. These complications predominantly manifest during the electrolyte and fluid correction phase, suggesting a possible influence of the rehydration therapy administered to these patients [9].
Since, as previously mentioned, evidence regarding the treatment of NHD remains limited, we deemed it appropriate to conduct a study aimed at determining the incidence in our region, investigating potential risk factors to improve prevention of neonatal hypernatremic dehydration, and assessing whether there is a statistical association between complications occurrence and the rehydration regimen employed. The ultimate aim is to generate new hypotheses to guide future studies that enable the development of standardized management protocols for this condition.
PATIENTS AND METHODS
This is a retrospective cohort observational study of newborns diagnosed with HND. An initial descriptive analysis is conducted, followed by an analytical study evaluating potential risk factors for developing HND, as well as the statistical association between different rehydration methods, related complications, and the rate of serum sodium decline. The aim is to generate hypotheses for future research. Population values provided by the Spanish National Institute of Statistics (INE) are used as reference standards.
Patients were retrospectively collected from May 2015 to January 2023. The study is conducted in a level IIIA Neonatal Intensive Care Unit (NICU). Demographic, clinical, laboratory, therapeutic, and outcome data were obtained through medical chart review. Access to medical records was granted after obtaining written informed consent from the parents. The study was reviewed and approved by the Research Ethics Committee for Medicinal Products in the autonomous community of La Rioja (CEImLaR).
Inclusion criteria were age under 28 days at the time of admission, a diagnosis of HND (defined by clinical signs consistent with dehydration along with a plasma sodium level ≥150 mEq/L at admission), and requiring admission to the hospital’s NICU. Exclusion criteria included lack of signed parental consent, missing data on serum sodium at admission, missing data on rehydration method within the first 24 hours, and absence of follow-up data in the medical record for at least six months after admission.
Primary variables include venous serum sodium level at admission, initial rehydration route, rate of serum sodium decline, and occurrence of complications. Secondary variables include sex, mode of delivery, maternal primiparity, birth during the warm season, type of feeding, age in hours at discharge from the maternity ward and at NICU admission, weight loss at maternity discharge and at admission, patient origin at admission, reason for consultation, use of volume expansion in the emergency department, sodium concentration of IV fluids for those who received them, length of stay, and subsequent neurodevelopmental abnormalities.
Regarding data analysis, appropriate statistical methods are applied based on the assumptions of each model, and differences and odds ratios are estimated using generalized linear or logistic regression models. Statistical analysis is performed using SPSS® software. Quantitative variables following a normal distribution are described as mean ± standard deviation (SD), while non-normally distributed variables are described as median and interquartile range (IQR), or log-transformed when appropriate to correct distribution. Categorical variables are expressed as frequencies and percentages.
The significance level is set at p < 0.05. Results will be interpreted with caution, even when statistically significant, due to the potential for error introduced by the small sample size and multiple comparisons.
RESULTS
A total of 25 patients were included in our sample. The incidence of neonatal hypernatremic dehydration (HND) in our setting during the study period was 1.65 per 1,000 live births.
The mean maternal age was 33.24 years (SD ± 4.64), with 96% of mothers being primiparous. 64% of the newborns were delivered via spontaneous vaginal delivery, 20% were born during warm months (defined as May through September), and 72% were male. All patients (100%) received exclusive breastfeeding prior to admission. The mean weight loss at discharge from the maternity ward was 8.4% (SD ± 1.93).
We identified the following as risk factors for developing neonatal HND: being born to a primiparous mother (p = 0.007), exclusive breastfeeding (p < 0.001), and male sex (p = 0.036). Cesarean delivery was found to be a protective factor (p = 0.007). No statistically significant differences were observed based on whether birth occurred during the warm months.
The primary reason for consultation at our center was referral from a primary care facility by pediatric or nursing staff (36%), followed by parental decision to seek care at the emergency department due to somnolence (20%) or jaundice (12%).
The mean age at admission was 5.3 days (SD ± 3.05). The average weight loss at admission was 16% relative to birth weight. The mean venous sodium level at admission is unknown, as one patient had an undetectable value (>180 mEq/L). Descriptive data regarding the characteristics of newborns are summarized in Table 1.
Volume expansion was administered in the emergency department in 16% of cases, with a mean volume of 21.3 cc/kg (SD ± 13.12). Among these patients, the type of fluid administered varied: one patient received 0.9% normal saline, two patients received 10% dextrose solution, and one patient received two boluses—one with 10% dextrose and another with 0.9% saline.
The route of rehydration during the first 24 hours was exclusively enteral in 44% of cases, mixed (enteral and intravenous) in 36%, and exclusively intravenous in 20%.
Of the 14 patients who received intravenous fluid therapy (56% of the sample), the mean volume administered during the first 24 hours was 73.19 mL/kg/day (SD ±57.96), and the mean amount of intravenous sodium administered was 3.26 mEq/kg/day (SD ±4.59). Notably, 4 patients did not receive sodium in their fluids. The median sodium concentration per liter of fluid was 26.64 mEq/L (IQR ±48.26), with a minimum of 0 and a maximum of 154.44 mEq/L.
Laboratory monitoring at 24 hours to assess the rate of sodium decline was performed in 18 patients (72%). The median sodium decline rate in our sample was 0.529 mEq/L/h (IQR ±0.64), with one outlier patient exhibiting a decline rate of 1.667 mEq/L/h. A total of 44.4% of patients demonstrated a decline rate <0.5 mEq/L/h. Descriptive data regarding rehydration variables are summarized in Table 2.
The mean length of stay was 4.9 days (SD ±4.66). Sixteen percent of the sample experienced complications, with seizures being the most frequent (3 cases). The follow-up period after discharge ranged from 6 months to 8 years. No cases with subsequent neurodevelopmental impairment were detected.
In our sample, a direct linear relationship exists between weight loss and the degree of hypernatremia (p<0.001, R² 0.773). Patients who received initial volume expansion demonstrated a higher complication rate (p=0.007) (Figure 1), as did the group with a sodium decline rate ≥0.5 mEq/L/h compared to the group with decline <0.5 mEq/L/h (p=0.041).
We found no association between the rate of sodium decline and either the sodium concentration in the infused solution or the absolute amount of sodium infused. A direct linear relationship exists between the rate of serum sodium decline and the volume of intravenous fluid administered (p<0.001, R² 0.733), with higher per-kilogram infused volume being associated with a greater rate of serum sodium decline. In our sample, fluid therapy administration at a rate of 51.86 mL/kg/day corresponds to a sodium decline rate of 0.5 mEq/L/h, with higher rates observed when greater volumes are infused. However, additional factors may influence this value.
DISCUSSION
The incidence of hypernatremic dehydration during the study period in our community (1.65/1000 live births per year) was closely aligned with that described in the medical literature, ranging between 0.5-10.4/1000 live births [2,3].
The proportion of male newborns admitted to our hospital for hypernatremic dehydration was 72%, compared to 28% for females. This distribution differs significantly from that observed in the general neonatal population (p=0.036), with the proportion of affected males being 20.4% higher (95% CI: 1%–39%). Therefore, male sex may constitute a risk factor, a finding not previously described in the literature, although confirmation in subsequent studies is required. In accordance with previous studies, other identified risk factors include being born to a primiparous mother (p=0.007) and exclusive breastfeeding (p<0.001). Previously described risk factors such as low birth weight or prematurity were not confirmed in our cohort, likely due to closer monitoring of these newborns. Although we hypothesized that birth during warm months might constitute a risk factor, our data do not support this assumption. Conversely, cesarean delivery appears to have a protective effect against admission for hypernatremic dehydration (p=0.007), possibly related to prolonged hospital stay following delivery. There were also no significant differences between the mean maternal age in our sample (33.24 years) and the population mean maternal age in our community (32.51 years) [1,5,10,11].
Unlike other series where the primary reason for consultation was weight loss, the main reason for consultation leading to admission in our study was referral by another healthcare professional from the primary care center [4].
The most frequent complication during the rehydration phase was the occurrence of seizures, observed in 12% of cases, a percentage higher than that described in other series [3,12].
In our sample, initial volume expansion was associated with a higher complication rate (p=0.007), contrary to findings in other series. However, it is possible that the initial clinical and laboratory severity of these patients was greater and acts as a confounding factor. Studies with stronger evidence are needed to assess the association between initial volume expansion and the occurrence of complications, as it may be necessary to restrict its use, limiting it to cases of shock and administering the minimum amount required [13].
Existing protocols, such as those from the Mexican Academy of Pediatrics and the United Kingdom's National Institute for Health and Care Excellence guidelines, recommend fluid therapy with higher tonicity than the mean used in our sample (26.64 mEq/L of sodium). Given the recommended sodium requirements in the neonatal period of 3 mEq/kg/day, the sodium intake recommended in the aforementioned guidelines may be excessive. The apparent lack of association between sodium decline rate and either the concentration or absolute amount of sodium infused in our study suggests that fluid volume may be more influential than sodium content in determining correction speed, though this hypothesis requires validation in controlled settings. The observed relationship between intravenous fluid volume and sodium decline rate (R² 0.733) suggests a potential association that warrants further investigation in larger cohorts. Our preliminary finding that approximately 51.86 mL/kg/day may correspond to the recommended sodium decline rate of 0.5 mEq/L/h could serve as a hypothesis-generating observation for future prospective studies. However, given our limited sample size and the retrospective nature of our analysis, this threshold should be considered exploratory rather than definitive. These preliminary observations align with current clinical approaches emphasizing fluid volume management in hypernatremic dehydration, but the complex pathophysiology involved and multiple potential confounding variables underscore the need for larger, prospective studies to establish evidence-based protocols. Our findings should be interpreted as generating hypotheses for future research rather than providing definitive clinical guidance [13-15].
In our region, the first well-child check occurs at 15 days of life. Given that most admissions in our study corresponded to infants referred by healthcare professionals, that weight loss correlated with the degree of hypernatremia, and that the mean age at admission was 5.3 days, the importance of earlier follow-up becomes evident. We propose implementing a follow-up visit at 4 days of life or 2 days after hospital discharge for high-risk neonates (defined as weight loss ≥8% at discharge combined with exclusive breastfeeding) to prevent or enable early detection of hypernatremic dehydration. However, further studies are needed to establish both the necessity of earlier monitoring and the effectiveness of the proposed timeframes according to individual newborn characteristics.
This study has important limitations: small sample size, single-center and retrospective design, and risk of false positives due to multiple comparisons. However, as an exploratory study aimed at generating hypotheses, these limitations do not preclude achieving the stated objectives.
Based on our findings, we suggest the following hypotheses for future studies: first, to evaluate whether restricting initial volume expansion to cases of shock reduces complications; and second, to determine whether, when oral rehydration is not feasible, initiating slow intravenous rehydration (even slightly below the neonate’s basal fluid requirements), with gradual progression and early combination with enteral feeding, improves neurological outcomes by avoiding serum sodium decreases >0.5 mEq/L/hour.
Further studies are needed to provide evidence for the management of neonatal hypernatremic dehydration and to enable the development of standardized treatment protocols.
STATEMENTS
Ethics Approval Statement
Reviewed and approved by the Clinical Research Ethics Committee of La Rioja (CEImLAR) on September 19, 2023, under reference CEImLAR PI 700. CEImLAR adheres to Good Clinical Practice (CPMP/ICH/135/95) standards and complies with current legislation governing its operations. The evaluation meeting fulfilled the requirements established in the applicable legislation of La Rioja (Spain) to validate the Ethics Committee's decision.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors declare that they have no conflict of interest.
REFERENCES
- López D, Alonso M, Ramos JM, Cordón AM, Sánchez Tamayo T, Urda L. (2018). Severe neonatal hypernatremic dehydration due to failure to establish breastfeeding: a study of incidence and associated factors. [Deshidratación hipernatrémica grave neonatal por fallo en la instauración de la lactancia materna: estudio de incidencia y factores asociados]. Rev Pediatr Aten Primaria; 20(79):229-235.
- Álvarez CFH, Robledo JFG, López AV. (2014). Clinical course of hepernatremic dehydration in newborns. [Curso clínico de la deshidratación hepernatrémica en recién nacidos]. Arch Investig Matern Infant. 6:52-60.
- Peñalver O, Gisbert J, Casero J, Bernal A, Oltra M, Tomás M. (2004). Hypernatremic dehydration associated with breastfeeding. [Deshidratación hipernatrémica asociada a lactancia maternal]. Anales de Pediatría (Barc). 61:340-343.
- Moritz ML, Manole MD, Bogen DL, Ayus JC. (2005). Breast-feeding-associated hypernatremia: are we missing the diagnosis? Pediatrics. 116(3):343-347.
- Asturizaga A, Mazzi E. (2011). Hipernatremia neonatal: factores de riesgo. Revista chilena de pediatría. 82(2):150-151.
- Ben-Shalom E, Toker O, Schwartz S. (2016) Hypernatremic dehydration in Young children: is there a solution?. The Israel Medical Association journal. 18(2):95-99. 31.
- Rincón DA. (2007). A quick formula for correcting sodium disorders. [Fórmula rápida para la corrección de los trastornos del sodio]. Revista de la Facultad de Medicina de Colombia [Internet]. [Consultado el 4 de Abril de 2025]. 55(1):63-65.
- Molteni KH. (1994). Initial management of hypernatremic dehydrationin the breastfed infant. Clinical Pediatrics. (Phila). 33(12):731-740.
- Unal S, Arhan E, Kara N, Uncu N, Aliefendioglu D. (2008). Breastfeeding associated hypernatremia: Retrospective analysis of 169 term newborns. Pediatrics international. 50(1):29-34.
- Instituto nacional de estadística. Population and average age by sex and country of birth [Población y Edad Media por país de nacimiento y sexo]. [Internet].
- Insituto Nacional de Estadística. Middle Ages to Maternity by birth order by autonomous community, according to the mother's nationality (Spanish/foreign). [Edad Media a la Maternidad por orden del nacimiento por comunidad autónoma, según nacionalidad (española/extranjera) de la madre]. [Internet].
- González LG, Carrera L, Arias RP, Romero MC, Suárez M, Ibañez A. et al. (2016). Hypernatremic dehydration associated with breast-feeding. [Deshidratación hipernatrémica asociada a la alimentación con lactancia materna en el periodo neonatal]. Anales de Pediatría. 74:261-265.
- Jonguitud- A, Calvillo S, Ruiz E, Olvera G. (2015). Management protocol for neonatal hypernatremic dehydration. [Protocolo de manejo en deshidratación hipernatrémica neonatal]. Elsevier. 29:2:65-69.
- Schwaderer AL, Schwartz GJ. (2005). Treating hypernatremic dehydration. Pediatrics in Review. 26(4):148-150.
- Grupo de trabajo de hidratación y trastornos electrolíticos SEUP. (2019). Manual para el diagnóstico y tratamiento de la deshidratación y los trastornos electrolíticos en urgencias de pediatría.. [Internet].
APPENDICES
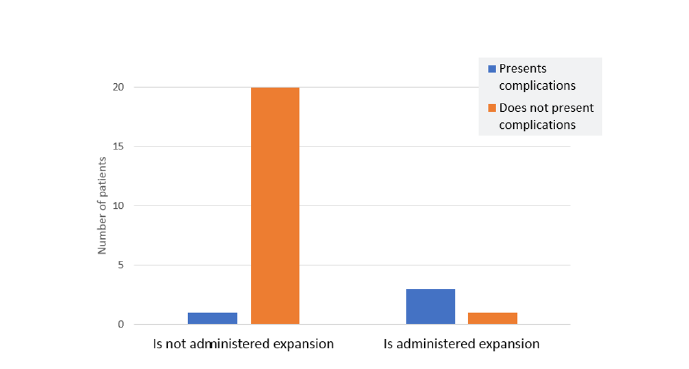
Figure 1: Count of patients presenting complications based on whether volume expansion is administered
Table 1: Descriptive Statistics (perinatal data, at admission, during treatment, and after treatment)
|
Variables (unit of measurement) |
Mean/median/% |
Min-max |
|
Maternal age (years) |
Mean 33,24 (SD 4,64) |
11-45 |
|
Primiparous |
96% |
|
|
Vaginal delivery |
64% |
|
|
Male sex |
72% |
|
|
Birth in warm months (May-September) |
20% |
|
|
Exclusive breatfeeding prior to admission |
100% |
|
|
High weight loss at maternity (% weight) |
Mean 8,4 (SD 1.93) |
4,8-12.6 |
|
Age at admission (days) |
Mean 5,3 (SD 3,056) |
2,5-15 |
|
Weight loss at admission (% weight) |
Mean 16 (SD 6,25) |
9,3-35 |
|
Length of stay (days) |
Mean 4.9 (SD 4,659) |
1-22 |
|
Presents complications |
16% |
|
|
Presents neurodevelopmental impairment |
0% |
|
Table 2: Descriptive Statistics: Variables on Rehydration
|
Variables (unit of measurement) |
Mean/median/% |
Min-max |
|
|
Receives initial volume expansion |
16% |
||
|
Amount of volume expansion administered (cc/kg) |
Mean 21, 3 (SD 13,12) |
10-40 |
|
|
Rehydration route in the first 24 hours |
Exclusive enteral |
44% |
|
|
Mixed (IV +enteral) |
36% |
||
|
Exclusive intravenous |
20% |
||
|
Volume of fluid therapy IV in the first 24 h. (cc/kg/24h) |
Mean 73,19 (SD 57,96) |
11, 63-221, 14 |
|
|
Amount of sodium in the first 24 hours (mEq/kg/24h) |
Mean 3,26 (SD 4,59) |
0-15, 18 |
|
|
Sodium concentration in fluid therapy in first 24 h (mEq/L) |
Median 26,64 (IQR 48,264) |
0-154 |
|
|
Rate of sodium decrease (mEq/L/) |
Median 0,529 (IQR 0,646) |
0, 196-1, 667 |
|
|
% of patients with sodium dcrease ≤.5 mEq/L/h |
44.44% |
||
 Abstract
Abstract  PDF
PDF