Current Issue
Impact of Fetal Growth Restriction on Neonatal Outcomes, Neurodevelopment and Growth - Experience of a Tertiary Center
Inês Aires Martins1*, Joana Carvalho Queirós1, Inês Carvalho1, Dora Sousa1, Ana Cunha2, Luís Guedes-Martins2, Teresa Borges3, Ana Cristina Barros1, Marta Nascimento1, Liliana Carvalho Teixeira1, Sara Domingues1, Carmen Carvalho1, Ana Novo1
1Department of Neonatology, Centro Materno-Infantil do Norte Albino Aroso, Unidade Local de Saúde de Santo António, Portugal
2Department of Obstetrics, Fetal Medicine Center, Centro Materno-Infantil do Norte Albino Aroso, ULS Santo António, Portugal
3Unit of Pediatric Endocrinology, Department of Pediatrics, Centro Materno-Infantil do Norte Albino Aroso, Unidade Local de Saúde de Santo António, Portugal
*Corresponding author: Inês Aires Martins, Department of Neonatology, Centro Materno-Infantil do Norte Albino Aroso, Unidade Local de Saúde de Santo António, Portugal, Phone: +351 222077500, E-mail: [email protected]
Received Date: February 05, 2026
Publication Date: March 15, 2026
Citation: Martins IA, et al. (2026). Impact of Fetal Growth Restriction on Neonatal Outcomes, Neurodevelopment and Growth - Experience of a Tertiary Center. Neonatal. 7(1):30.
Copyright: Martins IA, et al. © (2026).
ABSTRACT
Introduction: Fetal growth restriction (FGR) is associated with increased perinatal morbidity and adverse long-term neurodevelopmental and growth outcomes. Study Design: Retrospective cohort study of 789 newborns with FGR delivered at a level III hospital (2015–2019), evaluating neonatal outcomes, neurodevelopment at 24–36 months, and growth beyond 4 years. Results: Early-onset FGR accounted for 25.5% of cases. Prematurity, early-onset FGR, abnormal Doppler findings, and lower estimated fetal weight independently predicted neonatal morbidity and NICU admission. Neurodevelopmental impairment at 24-36 months was identified in 34.2% of assessed children. At follow-up beyond 4 years of age, 6.3% had persistent short stature. Growth hormone therapy showed favorable response. Conclusion: FGR is a heterogeneous condition with outcomes strongly determined by timing of onset, Doppler abnormalities, and growth phenotype. Early-onset FGR, abnormal Doppler findings, and symmetrical FGR, define a high-risk subgroup with significant neonatal, neurodevelopmental, and growth consequences, supporting individualized surveillance and targeted long-term follow-up.
Keywords: Fetal Growth Restriction, Neonatal Outcome, Neurodevelopment Outcome, Nutrition/Growth, Small-For-Gestational-Age
INTRODUCTION
Fetal growth restriction (FGR) affects up to 10% of pregnancies and reflects the failure of the fetus to reach its genetically determined growth potential [1-3]. International guidelines recommend a multimodal diagnostic approach integrating fetal biometry, Doppler parameters (umbilical artery, middle cerebral artery, cerebroplacental ratio), and markers of placental dysfunction (reflected by reduced placental weight) [1-3]. Early-onset FGR is typically associated with placental insufficiency and umbilical artery Doppler abnormalities, whereas late-onset FGR more often presents with cerebral redistribution as an adaptive response to chronic hypoxemia [1]. Placental insufficiency and Doppler abnormalities are strongly linked to adverse perinatal outcomes, including preterm birth and increased neonatal intensive care unit (NICU) admission [2,4]. Birthweight is a critical determinant of neonatal morbidity, particularly hypoglycemia, thermoregulatory instability, feeding difficulties, jaundice, and sepsis [6-8]. Reported rates of neonatal complications are also particularly high in early-onset or severe FGR, with NICU admission ranging from 45 to 70%, and perinatal mortality approaching 10-12% [5-7].
The consequences of FGR extend beyond the neonatal period. Neurodevelopmental impairment is well documented, with international cohorts reporting abnormal IQ scores in up to 35% and motor difficulties in 38% of children with early-onset FGR. Risk factors for adverse neurodevelopment include lower birthweight, male sex, neonatal morbidity, and greater severity of FGR [8,9]. Children with early-onset FGR and persistent Doppler abnormalities exhibit the highest risk of cognitive and motor deficits persisting into early childhood [8-11].
Growth trajectories are also altered. Although 85-90% of children born with FGR or small for gestational age (SGA) achieve catch-up growth by age 2, approximately 10-15% remain with short stature and impaired growth velocity [12-14]. Persistent short stature warrants evaluation for growth hormone (GH) therapy, and early initiation is associated with improved height outcomes and favourable safety profiles [12,15,16].
In summary, FGR is a multifactorial condition with both acute and chronic consequences. Antenatal parameters and birthweight are key for risk stratification, while neonatal morbidity, growth patterns, and neurodevelopmental outcomes guide long-term follow-up. Identifying predictive factors for adverse outcomes is essential to optimize surveillance and guide early interventions [1,2,12,13].
METHODS
Study Design and Sample
A retrospective cohort study was conducted, including newborns diagnosed with FGR delivered at a level III Portuguese hospital between January 2015 and December 2019. Exclusion criteria included stillbirths and major congenital anomalies identified prenatally.
Data Collection and Variables Definition
Data collection was performed through review of electronic medical records. Missing values were addressed using complete-case analysis for each outcome. Most variables had less than 10% missing data, and higher proportions were taken into consideration in the interpretations of results. Loss to follow-up was documented, and children excluded from analyses due to genetic alterations or incomplete data were noted.
Three follow-up periods were analysed: the neonatal period, neurodevelopmental evaluation at 24-36 months, and growth parameters beyond 48 months of age.
FGR was defined as a fetal weight below the 10th percentile for gestational age on antenatal ultrasound and classified according to gestational age at diagnosis: early-onset FGR if detected at or before 32 weeks, and late-onset FGR if diagnosed after 32 weeks. Placental characteristic, including weight and Doppler flow parameters were recorded when available. Prenatal placental function was evaluated using Doppler flow parameters, with abnormal Doppler defined as absent or reversed end-diastolic flow in the umbilical artery or a cerebroplacental ratio below the 5th percentile. Placental weight was recorded postnatally, and low placental weight was defined as below the 10th percentile for gestational age.
Anthropometric measures at birth were expressed according to Fenton growth curves. Infants were classified as light for gestational age (LGA) or SGA when measures were below the 3rd percentile for weight or length, respectively. Symmetrical LGA was considered when both weight and head circumference were below the 3rd percentile. Screening for cytomegalovirus (CMV) infection in urine using molecular methods was performed according to clinical indication. Genetic investigations, including chromosomal microarray, karyotyping, or targeted molecular testing, were undertaken when clinically justified after evaluation by a medical geneticist.
Neurodevelopment was evaluated in all children using the Mary Sheridan Developmental Assessment Scale, with those showing suspected delays undergoing formal assessment using the Ruth Griffiths Mental Development Scales between 24 and 36 months of age. Neurodevelopmental assessment below the population mean was defined as a score below 85 in any domain.
Growth outcomes beyond 48 months were assessed using height standard deviation scores (SDS) according to World Health Organization (WHO) growth charts. Short stature was defined as height below -2.5 SDS for age and sex. Data on initiation and response to growth hormone therapy were collected, with clinical response defined as an annual increase in height SDS ≥0.3-0.5 or a height velocity above -1 SDS for age and sex [17,18].
Statistical Analysis
Statistical analysis was performed with IBM® SPSS® Statistics version 28.0. Categorical variables were expressed as frequencies (n) and percentages (%), and continuous variables as means (M) and standard deviations (SD), or medians (Mdn) and interquartile ranges (IQR) for variables with skewed distributions. Normality was assessed using the Shapiro-Wilk test. Chi-square or Fisher’s exact test for categorical variables, and independent samples t-tests or Mann-Whitney U test for continuous variables were used. Logistic regression was performed to explore predictive factors for different outcomes. All tests were two-sided, and statistical significance was defined as p<0.05.
RESULTS
Study Population
A total of 789 newborns with FGR were included, accounting for 4.6% of births during the study period (n=17152). Early-onset FGR accounted for approximately one quarter of cases (n=201, 25.5%). The median EFW was 2110g (IQR 1639-2304), corresponding to the 6th percentile (IQR 2-7). Abnormal placental Doppler flows were documented in 27.1% (n=214), and low placental weight was observed in 37.9% (n=299), with a median of 326g (IQR 272-387), corresponding to the 5th percentile (IQR 3-9).
Perinatal and Neonatal Characteristics
The median gestational age was 37 weeks (IQR 35-38), with 31.8% born preterm (n=251). The median birthweight was 2390g (IQR 1765-2490), corresponding to the 9th percentile (IQR 3-15). More than half of the newborns were below the 10th percentile for birthweight (n=422, 53.5%), and one fifth below the 3rd percentile, thereby classified as LGA (n=171, 21.7%). Median birth length was 45.5cm (IQR 41-46), with SGA status observed in 22.9% (n=181). Symmetrical LGA was identified in 3.2% (n=25).
LGA status was significantly more common among preterm newborns (OR 1.569, p=0.012), those with low placental weight (OR 3.303, p<0.001), early-onset FGR (OR 0.497, p<0.001), and those requiring NICU admission (OR 3.043, p<0.001). Symmetrical LGA was associated with lower EFW (Cohen’s d=0.995, p=0.003), prematurity (OR 3.979, p<0.001), early-onset FGR (OR 0.079, p<0.001), low placental weight (OR 5.848, p<0.001), and NICU admission (OR 15.344, p<0.001).
These results are further characterized in Table 1 and Table 2.
Table 1: Perinatal and neonatal characteristics of the study population.
|
Characteristics |
Total cohort (n=789) |
|
Sex, n (%) Male Female |
368 (46.6) 421 (53.4) |
|
Fetal Growth Restriction, n (%) Early-onset FGR Late-onset FGR |
201 (25.5) 588 (74.5) |
|
Estimated Fetal Weight Median EFW, g (IQR) Median EFW percentile (IQR) |
2110g (1639-2304) 6 (2-7) |
|
Placental Doppler Flow, n (%) Normal Doppler flow Abnormal Doppler flow |
214 (27.1) 575 (72.9) |
|
Placental Weight Median placental weight, g (IQR) Median placental weight percentile (IQR) Placental weight <P10, n (%) |
326 (272-387) 5 (3-9) 299 (37.9) |
|
Gestational Age Median gestational age, weeks (IQR) Preterm, n (%) |
37 (35-38) 251 (31.8) |
|
Anthropometric Measures at Birth Median birthweight, g (IQR) Median birthweight percentile (IQR) Birthweight <P3, n (%) Birthweight <P10, n (%)
Median length, cm (IQR) Median length percentile (IQR) Length <P3, n (%)
Median head circumference, cm (IQR) Median head circumference percentile (IQR) Head circumference <P3, n (%)
Symmetrical light for gestational age |
2390 (1765-2490) 9 (3-15) 171 (21.7) 422 (53.5)
45.5 (41-46) 11 (3-19) 181 (22.9)
32.5 (30-33) 25 (9-42) 70 (8.9)
25 (3.2) |
Table 2: Perinatal characteristics according to birthweight phenotype.
|
VARIABLE |
LGA |
NOT- LGA |
OR (CI 95%) |
p-value |
SYMMETRICAL LGA |
NOT-SYMMETRICAL LGA |
OR (CI 95%) |
p-value |
|
Early-onset FGR |
31.3% |
18.5% |
0.497 (0.3-0.7) |
<0.001 |
10.0% |
0.9% |
0.079 (0.0-0.2) |
<0.001 |
|
Abnormal Doppler flow |
28.4% |
27.8% |
- |
0.644 |
50.0% |
27.3% |
- |
0.317 |
|
Low placental weight |
36.8% |
15.0% |
3.303 (2.2-5.0) |
<0.001 |
6.7% |
1.2% |
5.848 (1.7-19) |
0.001 |
|
Prematurity |
27.1% |
19.1% |
1.569 (1.1-2.2) |
0.012 |
6.4% |
1.7% |
3.979 (1.7-9.1) |
<0.001 |
|
CMV infection |
1.2% |
0.8% |
- |
0.257 |
0.0% |
0.8% |
- |
0.447 |
|
NICU admission |
36.7% |
16.0% |
3.043 (2.1-4.3) |
<0.001 |
9.8% |
0.7% |
15.344 (5.2-45)) |
<0.001 |
Acute Neonatal Outcomes
More than one quarter of newborns required NICU admission (n=215, 27.2%). Acute complications were recorded in 14.4% (n=114), the most frequent being hypoglycaemia (n=59, 7.5%), thrombocytopenia (n=49, 6.2%), polycythaemia (n=5, 0.6%) and necrotizing enterocolitis (n=1, 0.1%).
CMV screening was performed in 13% of newborns (n=101), with 5 positive results (5%). Among the higher-risk group of infants with early-onset FGR or symmetrical LGA (n=206), none tested positive. Of those high-risk infants who were not tested (n=157, 76%), 100 (64%) had documented “non-immune” maternal CMV serology in the third trimester.
Genetic investigations were conducted in 5.6% (n=44), and alterations were identified in 1.4% (n=11). These included chromosomal syndromes (trisomy 21, chromosomal anomaly involving chromosome 16, 1q21.1 microdeletion syndrome), monogenic syndromes with identified mutations (GNAS, SHOX, ZMYM3, MYT1L), and clinically recognized syndromes (acrodisostosis type 1, Barth syndrome, cerebellar atrophy with short stature and lipodystrophy phenotype, and an unspecified polymalformative syndrome).
The overall neonatal mortality rate was 1.3% (n=10), corresponding to 4.7% of NICU admissions. Causes of death included severe neurological events (intraventricular haemorrhage with infarction, hypoxic-ischemic encephalopathy after cardiorespiratory arrest (n=2), refractory metabolic acidosis with suspected mitochondrial disease), respiratory causes (bilateral tension pneumothorax, massive pulmonary haemorrhage (n=2), refractory respiratory failure), refractory septic shock, and necrotizing enterocolitis with multiorgan dysfunction.
Independent predictors of acute neonatal complications were prematurity (OR 3.956, p<0.001), LGA (OR 2.502, p=0.001), lower EFW (OR 0.99, p<0.001) and lower Apgar score (OR 0.779, p=0.034) (Hosmer-Lemeshow: χ²(8)=2.985, p=0.935). NICU admission was independently predicted by prematurity (OR 19.571, p<0.001), symmetrical LGA (OR 9.947, p=0.006), LGA (OR 3.778, p<0.001), early-onset FGR (OR 2.628, p=0.001), lower EFW (OR 0.99, p<0.001), and lower Apgar scores (OR 0.438, p<0.001) (Hosmer-Lemeshow: χ²(8)=7.146, p=0.521). Although lower EFW was statistically associated with both acute complications and NICU admission, the effect size was small (OR 0.999). Abnormal placental Doppler flow was significantly more common among NICU-admitted newborns (OR 1.889, p<0.001). These results are further characterized in Table 3.
Table 3: Multivariable logistic regression analyses for neonatal outcomes.
A. Neonatal complications.
|
VARIABLE |
aOR |
CI (95%) |
p-value |
|
Prematurity |
3.956 |
2.404-7.672 |
<0.001 |
|
LGA |
2.502 |
1.439-4.349 |
0.001 |
|
EFW |
0.999 |
0.998-1.0 |
<0.001 |
|
Apgar Score 5’ |
0.779 |
0.619-0.982 |
0.034 |
B. NICU admission.
|
VARIABLE |
aOR |
CI (95%) |
p-value |
|
Prematurity |
19.571 |
10.337-37.056 |
<0.001 |
|
Symmetrical LGA |
9.947 |
1.909-51.848 |
0.006 |
|
LGA |
3.778 |
1.897-7.524 |
<0.001 |
|
Early-onset FGR |
2.628 |
1.456-4.744 |
0.001 |
|
EFW |
0.999 |
0.310-0.619 |
<0.001 |
|
Apgar Score 5’ |
0.438 |
0.998-0.999 |
<0.001 |
Neurodevelopmental Outcomes
Formal evaluation with the Ruth Griffiths Mental Development Scales were available for 108 children, accounting for 14.1% of the cohort, with a median age of 35 months (IQR 34–36).
Compared with children without follow-up data, those assessed did not differ significantly in sex distribution (p=0.121), presence of Doppler alterations (p=0.201), placental weight (p=0.377), or CMV positivity (p=0.771). However, assessed infants had a higher incidence of neonatal complications (66.9% vs. 15.6%, p<0.001), early-onset FGR (50.3% vs. 18.1%, p<0.001), lower birthweight percentiles (median 9.41 vs. 14.69, p<0.001), and were born at a lower gestational age (median 34.16 vs. 37.39 weeks, p<0.001), suggesting that the follow-up subset represented a slightly higher-risk population.
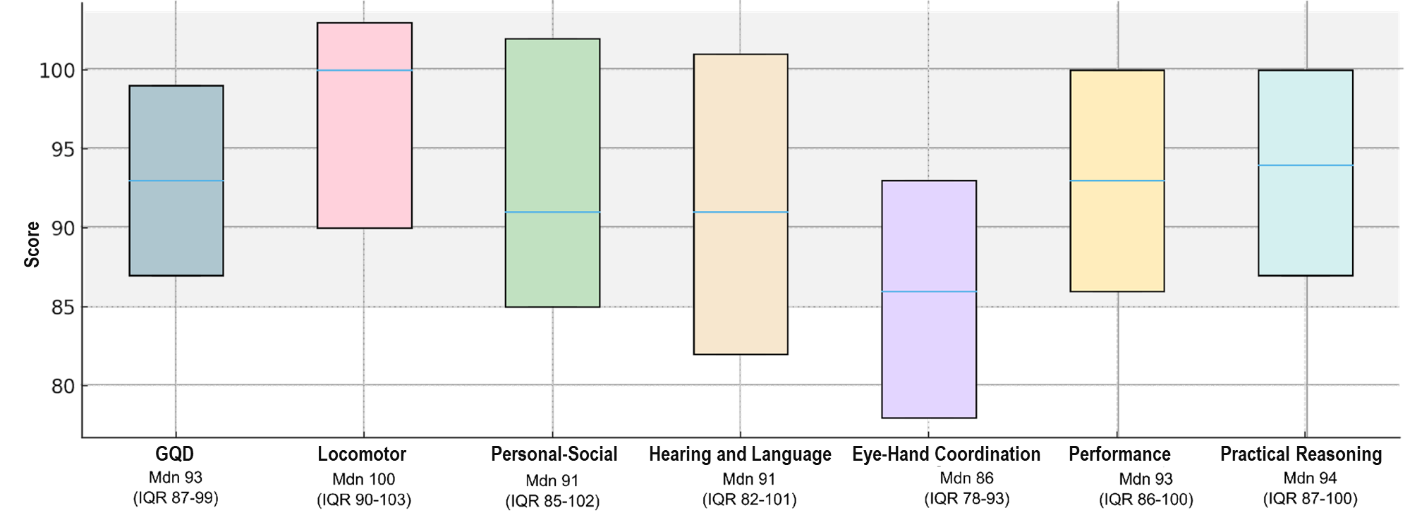
The median general quotient of development (GQD) was below the population mean in 34.2% (n=37). Oculomotor coordination and language were the most frequently affected domains, with 54.6% (n=59) and 48.1% (n=52) children, respectively, scoring below the expected range.
Children with abnormal Doppler findings had significantly lower GQD (p=0.012), as well as in specific domains, particularly locomotor (p=0.001), eye-hand coordination (p=0.011), and performance (p=0.017) scores. Prematurity was associated with lower global (p=0.046), personal-social (p=0.040), and language (p=0.005) scores. CMV infection was associated with impaired neurodevelopment across several domains: global (p=0.005), locomotor (p=0.019), personal-social (p=0.013), language (p=0.011), and performance (p=0.049). These results are represented in Figure 1 and Table 4.
NICU admission (OR 8.331, p=0.048), abnormal Doppler flows (OR 3.592, p=0.032), prematurity (OR 0.278, p=0.031), and symmetrical LGA (OR 0.111, p=0.014) were identified as independent predictors of altered neurodevelopmental outcomes (Hosmer-Lemeshow: χ²(4)=1.784, p=0.775), as depicted in Table 4B.
Figure 1: Neurodevelopmental outcomes according to the Ruth Griffiths Mental Development Scales (n=108).
Table 4 Shows characterization and multivariable logistic regression analysis for neurodevelopment outcomes according to the Ruth Griffiths Mental Development Scales GQD score (n=108).
Table 4A: Characterization of neurodevelopment outcomes.
|
VARIABLE |
LOWER THAN AVERAGE |
NORMAL |
OR |
p-value |
|
GQD SCORE |
GQD SCORE |
(CI 95%) |
||
|
Early-onset FGR |
58.50% |
47.30% |
- |
0.587 |
|
Abnormal Doppler flow |
46.70% |
27.30% |
2.333 |
0.012 |
|
(0.972-5.599) |
||||
|
Low placental weight |
56.60% |
43.40% |
- |
0.576 |
|
Prematurity |
46.20% |
28.40% |
0.463 |
0.046 |
|
(0.186-1.149) |
||||
|
CMV infection |
3.80% |
1.30% |
2.537 |
0.005 |
|
(1.972-4.440) |
||||
|
NICU admission |
67.90% |
55.90% |
- |
0.62 |
|
LGA |
35.80% |
28.40% |
- |
0.388 |
|
Symmetrical LGA |
36.20% |
18.40% |
1.43 |
0.047 |
|
(1.132-4.344) |
Table 4B: Multivariable logistic regression analysis for neurodevelopment outcomes.
|
VARIABLE |
aOR |
CI (95%) |
p-value |
|
NICU admission |
8.331 |
0.979-70.882 |
0.048 |
|
Abnormal Doppler flow |
3.592 |
1.301-9.917 |
0.032 |
|
Prematurity |
0.278 |
0.278-0.891 |
0.031 |
|
Symmetrical LGA |
0.111 |
0.684-0.734 |
0.014 |
Growth Parameters Assessment
Growth data after 48 months were available for 682 children (86.4%), at a median age of 61 months (IQR 53–63). Median height corresponded to the 42nd percentile (IQR 18–70), with 6.3% (n=43) classified as having short stature. Height at follow-up correlated with birth length (Spearman’s p=0.160, p<0.001) and EFW (Spearman’s p=0.108, p=0.005).
Short stature was more frequent among preterm infants (OR 1.861, p=0.049), SGA (OR 3.601, p<0.001), LGA (OR 2.417, p=0.006), symmetrical LGA (OR 3.583, p<0.001), early-onset FGR (OR 0.297, p<0.001), and those requiring NICU admission (OR 2.253, p=0.010). Logistic regression confirmed early-onset FGR (OR 15.60, p=0.015) and SGA (OR 5.509, p=0.035) as independent predictors of short stature beyond 4 years (Hosmer-Lemeshow: χ²(3)=0.242, p=0.971), as seen in Table 5.
Growth Hormone Therapy
Among LGA/SGA children who later presented with short stature (n=18), 10 (56%) were referred to Paediatric Endocrinology, the remaining being lost to follow-up. GH therapy was initiated in 6 children (60% of those referred).
Treatment began at a median age of 57 months (IQR 52-84), with a median baseline height SDS of -2.46 (IQR -3.02 to -2.31) and median growth velocity SDS of -1.21 (IQR -1.35 to -0.91). After two years, 4 out of 5 patients with complete follow-up (1 patient had less than 12 months of therapy) met the established response criteria, with improved median height SDS (-2.05, IQR -2.91 to -1.71) and growth velocity SDS (0.00, IQR -0.67 to +0.14). Early-onset FGR (OR 7.829, p=0.045) and SGA (OR 9.427, p=0.047) were identified as independent predictors for GH therapy initiation (Hosmer-Lemeshow: χ²(8)=0.850, p=0.971), as seen in Table 5C.
Table 5 Shows characterization and multivariable logistic regression analysis for growth outcomes (n=682).
Table 5A: Growth outcomes.
|
VARIABLE |
SHORT STATURE |
NOT- |
OR |
p-value |
|
SHORT STATURE |
(CI 95%) |
|||
|
Early-onset FGR |
13.00% |
4.30% |
0.297 |
<0.001 |
|
(0.159-0.557) |
||||
|
Abnormal Doppler flow |
43.20% |
27.00% |
- |
0.681 |
|
Low placental weight |
50.00% |
37.30% |
- |
0.768 |
|
Prematurity |
9.00% |
5.10% |
1.861 |
0.049 |
|
(0.996-3.478) |
||||
|
CMV infection |
0.00% |
0.80% |
- |
0.435 |
|
NICU admission |
10.40% |
4.90% |
2.253 |
0.01 |
|
(1.197-4.240) |
||||
|
LGA |
11.30% |
5.00% |
2.417 |
0.006 |
|
(1.264-4.623) |
||||
|
Symmetrical LGA |
38.60% |
14.50% |
3.583 |
<0.001 |
|
(2.384-8.196) |
||||
|
SGA |
13.70% |
4.20% |
3.601 |
<0.001 |
|
(1.922-6.748) |
|
VARIABLE |
aOR |
CI (95%) |
p-value |
|
Early-onset FGR |
15.6 |
1.721-141.371 |
0.015 |
|
SGA |
5.509 |
1.130-26.867 |
0.035 |
Table 5C: Growth hormone therapy.
|
VARIABLE |
aOR |
CI (95%) |
p-value |
|
Early-onset FGR |
7.829 |
0.855-71.693 |
0.045 |
DISCUSSION
This large single-center cohort provides a comprehensive overview of perinatal and early-childhood outcomes in pregnancies affected by FGR. Consistent with recent systematic reviews and multicenter studies, early-onset FGR, prematurity, abnormal placental Doppler flows, and symmetrical LGA status emerged as the strongest independent predictors of adverse outcomes [1,2].
Prematurity was the dominant determinant of NICU admission, while early-onset FGR and Doppler abnormalities contributed substantially to acute neonatal morbidity. These associations mirror published data showing that early-onset FGR carries a markedly higher risk of perinatal death and severe neonatal complications [1,2,5,19]. The observed rates of NICU admission and acute morbidity in this study fall within the ranges reported in recent international cohorts, supporting the external validity of our findings [5,6].
Neurodevelopmental outcomes also align with the literature. Children with early-onset FGR, prematurity, and abnormal Doppler findings demonstrated lower cognitive, motor, and language scores at 24-36 months, consistent with prior meta-analyses and prospective cohorts [10,20,21]. Formal neurodevelopmental assessment was available in only 14.1% of the cohort, representing an important limitation. However, this reflects routine clinical practice, in which formal testing using the Ruth Griffiths Mental Development Scales is reserved for children with suspected developmental concerns following initial screening. As a result, the assessed subgroup represents a higher-risk population, characterized by lower gestational age, lower birthweight, and a greater burden of neonatal morbidity. This referral-based follow-up strategy introduces a potential selection bias, limiting the ability to estimate the true prevalence of neurodevelopmental impairment in the overall FGR population. While the associations identified between abnormal Doppler findings, NICU admission, prematurity, and adverse neurodevelopmental outcomes are biologically plausible and consistent with previous literature, the magnitude of observed impairment may be overrepresented in this subgroup.
Growth parameters assessment further highlights the heterogeneity of postnatal trajectories. While most children achieved catch-up growth by age four, a relevant proportion showed persistent short stature. This aligns with reports indicating that 10-15% of LGA/SGA children fail to achieve spontaneous catch-up growth [12,13]. Importantly, early-onset FGR and SGA were independent predictors of the need for GH therapy in our cohort, supporting prior evidence that the severity and timing of FGR are key determinants of treatment need [14]. Treatment response after two years was favourable in most children, echoing previously published data showing improved height trajectories with early GH initiation [12,15,16].
CONCLUSION
Overall, this study reinforces the substantial heterogeneity of FGR and highlights the prognostic value of combining gestational age at onset, fetal growth patterns, and Doppler findings. Early-onset FGR with Doppler abnormalities, especially symmetrical restriction, represents a high-risk phenotype. These findings are aligned with current evidence and support individualized surveillance and early intervention in high-risk subgroups. Future prospective, standardized follow-up studies are needed to refine prediction models and optimize long-term outcomes in children affected by FGR.
Strengths and Limitations
A major strength of this study is the integration of detailed perinatal and early-childhood follow-up data within a well-characterized cohort. This allowed a robust analysis of predictors from fetal assessment through long-term outcomes.
However, several limitations should be acknowledged. First, the single-center design may limit generalizability, although outcome rates were comparable to international cohorts. Second, despite the large initial sample, losses to follow-up, the exclusion of children with major genetic diagnoses, and the inherent variability in early-childhood assessments may limit precision in estimating long-term outcomes.
STATEMENTS
Statement of Ethics
This study was reviewed and approved by Santo António’s Ethics Committee - Instituto de Ciências Biomédicas Abel Salazar, approval number 2024.193 (160-CAC/152-CE). Given the retrospective nature of the study, the Ethics Committee deemed written informed consent unnecessary. This study was performed in accordance with the Declaration of Helsinki.
Conflict of Interest
The authors have no conflicts of interest to declare.
Data Availability
All data generated or analysed during this study are included in this article. Further enquiries can be directed to the corresponding author.
REFERENCES
- Lees CC, Romero R, Stampalija T, Dall’Asta A, DeVore GA, Prefumo F, et al. (2022). Clinical Opinion: The diagnosis and management of suspected fetal growth restriction: an evidence-based approach. American Journal of Obstetrics and Gynecology. 226(3):366-378.
- Society for Maternal-Fetal Medicine (SMFM), Martins JG, Biggio JR, Abuhamad A. (2020). Society for Maternal-Fetal Medicine Consult Series #52: Diagnosis and management of fetal growth restriction: (Replaces Clinical Guideline Number 3, April 2012). American Journal of Obstetrics and Gynecology. 223(4):B2–17.
- Lees C, Stampalija T, Hecher K. (2021). Diagnosis and management of fetal growth restriction: the ISUOG guideline and comparison with the SMFM guideline. Ultrasound in Obstetrics & Gynecology. 57(6):884-887.
- Kingdom JC, Audette MC, Hobson SR, Windrim RC, Morgen E. (2018). A placenta clinic approach to the diagnosis and management of fetal growth restriction. American Journal of Obstetrics and Gynecology. 218(2S):S803-S817.
- Pels A, Beune IM, van Wassenaer-Leemhuis AG, Limpens J, Ganzevoort W. (2020). Early-onset fetal growth restriction: A systematic review on mortality and morbidity. Acta obstetricia et gynecologica Scandinavica. 99(2):153-166.
- Tedyanto CP, Prasetyadi FOH, Dewi S, Noorlaksmiatmo H. (2025). Maternal factors and perinatal outcomes associated with early-onset versus late-onset fetal growth restriction: a meta-analysis. The Journal Of Maternal-Fetal & Neonatal Medicine. 38(1):2505774.
- Lingam I, Okell J, Maksym K, Spencer R, Peebles D, Buquis G, et al. (2023). Neonatal outcomes following early fetal growth restriction: a subgroup analysis of the EVERREST study. Archives of Disease in Childhood. Fetal and Neonatal Edition. 108(6):599-606.
- Pels A, Knaven OC, Wijnberg-Williams BJ, Eijsermans MJC, Mulder-de Tollenaer SM, Aarnoudse-Moens CSH, et al. (2019). Neurodevelopmental outcomes at five years after early-onset fetal growth restriction: Analyses in a Dutch subgroup participating in a European management trial. European Journal of Obstetrics, Gynecology, and Reproductive Biology. 234:63-70.
- Benítez Marín MJ, Blanco Elena JA, Marín Clavijo J, Jiménez López J, Lubián López DM, González Mesa E. (2022). Neurodevelopment Outcome in Children with Fetal Growth Restriction at Six Years of Age: A Retrospective Cohort Study. International Journal of Environmental Research and Public Health. 3:19(17):11043.
- Della Gatta AN, Aceti A, Spinedi SF, Martini S, Corvaglia L, Sansavini A, et al. (2023). Neurodevelopmental outcomes of very preterm infants born following early foetal growth restriction with absent end-diastolic umbilical flow. European Journal of Pediatrics. 182(10):4467-4476.
- Sun L, Lee FT, Milligan N, Zhu M, van Amerom JFP, Saini BS, et al. (2025). Neurodevelopment Among Infants With Late-Onset Fetal Growth Restriction. JAMA Network Open. 2:8(6):e2517360.
- Bamba V, Kanakatti Shankar R. (2022). Approach to the Patient: Safety of Growth Hormone Replacement in Children and Adolescents. The Journal of Clinical Endocrinology and Metabolism. 17:107(3):847-861.
- Ferrigno R, Savage MO, Cioffi D, Pellino V, Savanelli MC, Klain A. (2025). Effects of long-term treatment with recombinant growth hormone on growth outcome in children born small for gestational age: a systematic review. Reviews in Endocrine & Metabolic Disorders. 26(2):147-159.
- Lavi E, Shafrir A, Halloun R, Basel I, Eventov Friedman S, Abu-Libdeh A, et al. (2021). Eligibility for growth hormone therapy in children born small for gestational age is substantially lower than expected. Clinical Endocrinology. 95(2):308-314.
- Coutant R, Leheup B, Nicolino M, Salles JP. (2023). Growth Hormone Dose Modulation and Final Height in Short Children Born Small for Gestational Age: French Real-Life Data. Hormone Research in Pædiatrics. 96(5):495-508.
- Gli?ska M, Walczak M, Wikiera B, Pyr?ak B, Majcher A, Paluchowska M, et al. (2022). Response to Treatment with Recombinant Human Growth Hormone (rhGH) of Short Stature Children Born Too Small for Gestational Age (SGA) in Selected Centres in Poland. Journal of Clinical Medicine. 30;11(11):3096.
- Pozzobon G, Partenope C, Mora S, Garbetta G, Weber G, Barera G. (2019). Growth hormone therapy in children: predictive factors and short-term and long-term response criteria. Endocrine. 66(3):614-621.
- Dauber A, Phillip M, Ferran JM, Kelepouris N, Nedjatian N, Olsen AH, et al. (2024). Clinical Predictors of Good/Poor Response to Growth Hormone Treatment in Children with Idiopathic Short Stature. Hormone Research in Pædiatrics. 21:1-12.
- Leon-Martinez D, Lundsberg LS, Culhane J, Zhang J, Son M, et al. (2023). Fetal growth restriction and small for gestational age as predictors of neonatal morbidity: which growth nomogram to use? American Journal Of Obstetrics and Gynecology. 229(6):678.e1-678.e16.
- Olga L, Sovio U, Wong H, Smith G, Aiken C. (2023). Association between antenatal diagnosis of late fetal growth restriction and educational outcomes in mid-childhood: A UK prospective cohort study with long-term data linkage study. PLoS Medicine. 20(4):e1004225.
- Sacchi C, Marino C, Nosarti C, Vieno A, Visentin S, Simonelli A. (2020). Association of Intrauterine Growth Restriction and Small for Gestational Age Status With Childhood Cognitive Outcomes: A Systematic Review and Meta-analysis. JAMA Pediatrics. 174(8):772-781.
 Abstract
Abstract  PDF
PDF